 
Khan Academy is a nonprofit with the mission of providing a free, world-class education for anyone, anywhere. The first shows common element charges, while the second shows all the element charges for the first 45 elements (most common charges in bold). Learn for free about math, art, computer programming, economics, physics, chemistry, biology, medicine, finance, history, and more. They all have a similar electron configuration in their valence shells: a single s electron. Charts come from empirical data on the real behavior of elements, which may differ somewhere from the periodic table predictions. Their electron configurations (abbreviated for the larger atoms) are as follows, with the valence shell electron configuration highlighted: Electrons, electron configurations, and the valence shell electron configuration highlighted. For example, take the elements in the first column of the periodic table: H, Li, Na, K, Rb, and Cs. Valence electrons are of crucial importance because they lend deep insight into an element’s chemical properties: whether it is electronegative or electropositive in nature, or they indicate the bond. If we look at just the valence shell’s electron configuration, we find that in each column, the valence shell’s electron configuration is the same. Table of Contents (click to expand) Valence electrons are those electrons that reside in the outermost shell surrounding an atomic nucleus. To get the number of valence electrons just look at the numbers above the oxygen on the periodic table. In order to get the number of neutrons you take the atomic weight in this case 15.999916 and you subtract it by the number of protons (16-8). This means that oxygen has 8 protons and 8 electrons. (The inner electrons are called core electrons.) The valence electrons largely control the chemistry of an atom. Oxygen is the 8th element in the periodic table. The electrons in the highest-numbered shell, plus any electrons in the last unfilled subshell, are called valence electrons the highest-numbered shell is called the valence shell. 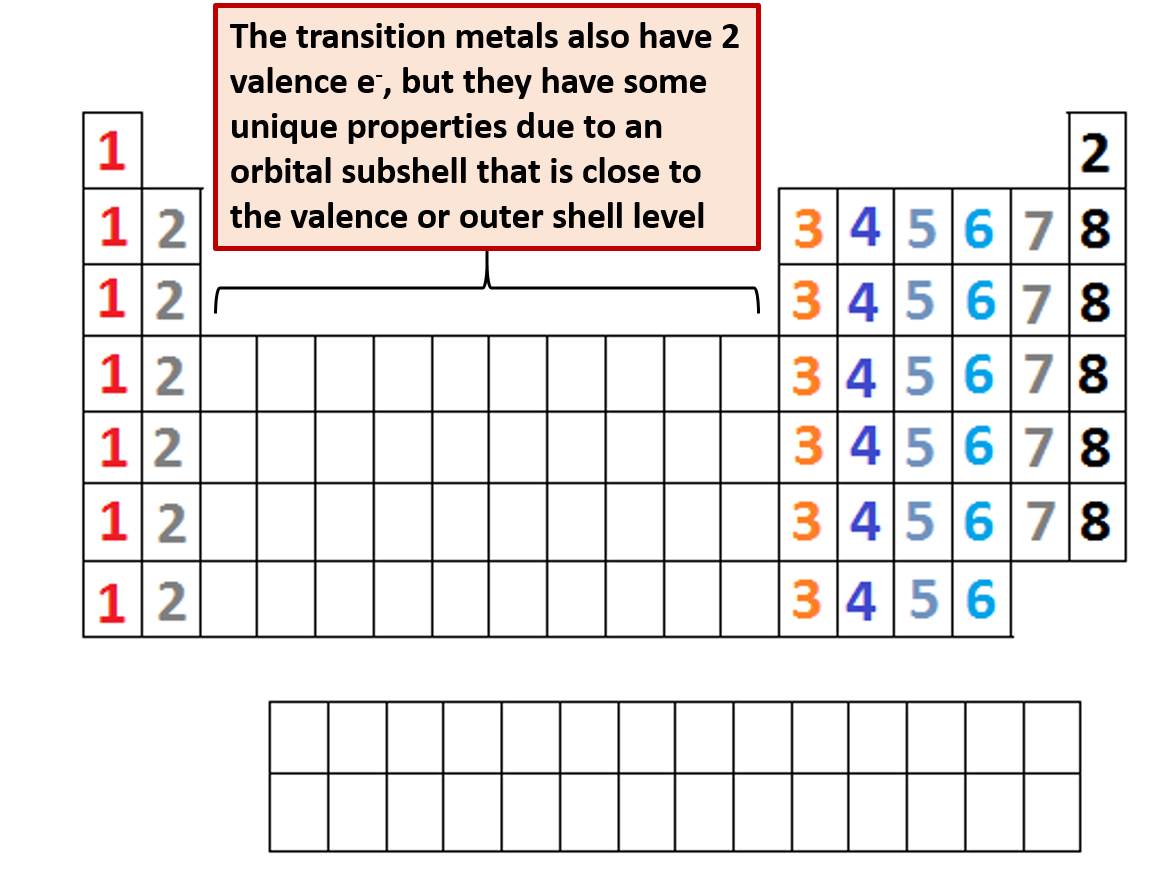 
The periodic table is separated into blocks depending on which subshell is being filled for the atoms that belong in that section. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
